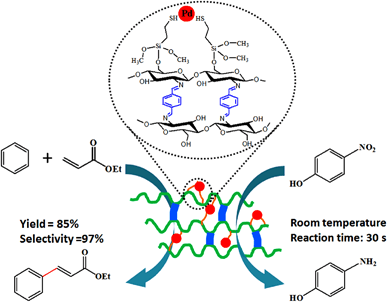
Thiol Functionalized Cross-Linked Chitosan Polymer Supporting Palladium for Oxidative Heck Reaction and Reduction of p-Nitrophenol - Catal. Lett. - X-MOL

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
Ten-fold boost of catalytic performance in thiol–yne click reaction enabled by a palladium diketonate complex with a hexafluoroacetylacetonate ligand - Catalysis Science & Technology (RSC Publishing)

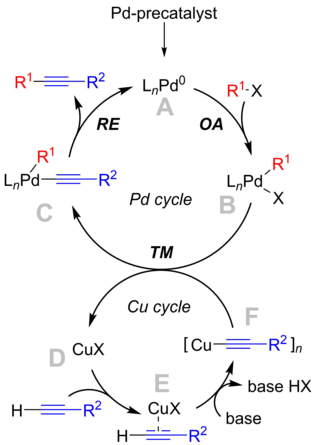
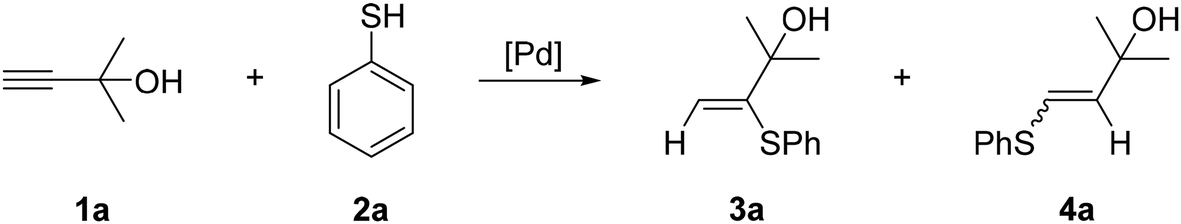
Alkyne Hydrothiolation Catalyzed by a Dichlorobis(aminophosphine) Complex of Palladium: Selective Formation of cis‐Configured Vinyl Thioethers - Gerber - 2012 - Chemistry – A European Journal - Wiley Online Library
Tuning the Catalytic Activity and Selectivity of Pd Nanoparticles Using Ligand-Modified Supports and Surfaces
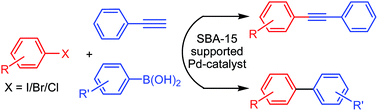
Highly active thiol-functionalized SBA-15 supported palladium catalyst for Sonogashira and Suzuki–Miyaura cross-coupling reactions - RSC Advances (RSC Publishing)

PDF) Palladium N-heterocyclic carbene catalyzed regioselective thiolation of 1-aryl-3-methyl-1H-pyrazol-5(4H)-ones using aryl thiols | Vishal Purohit - Academia.edu
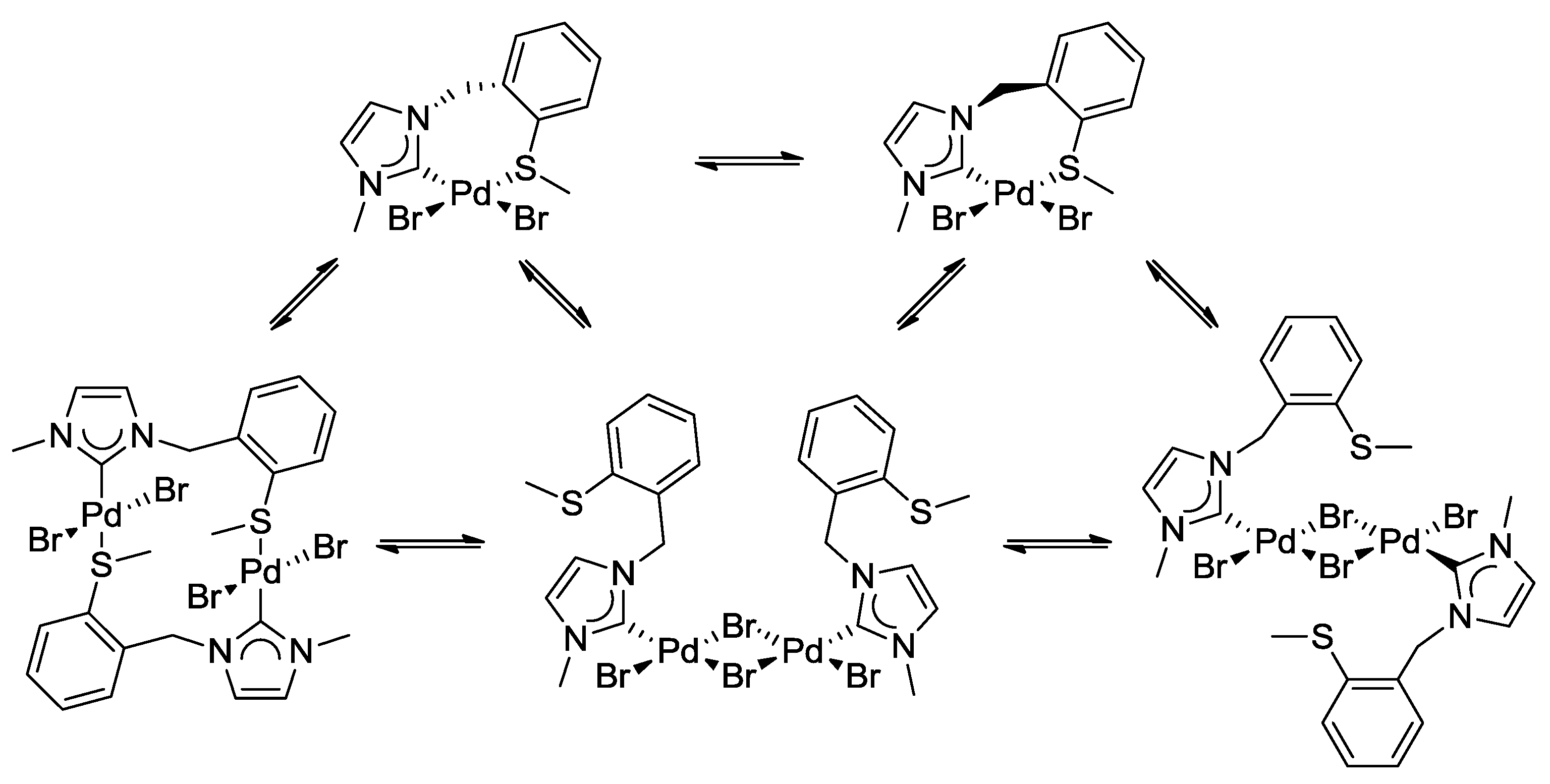
Molecules | Free Full-Text | Sulfur-Functionalized N-Heterocyclic Carbene Complexes of Pd(II): Syntheses, Structures and Catalytic Activities | HTML

Cellulose-supported N-heterocyclic carbene-palladium catalyst: synthesis and its applications in the Suzuki cross-coupling reaction. | Semantic Scholar

Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti‐Markovnikov‐Selective Hydrothiolation of Olefins - Sinha - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library

Catalysts | Free Full-Text | Oxidation of Thiol Using Ionic Liquid-Supported Organotelluride as a Recyclable Catalyst | HTML

Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect

Ten-fold boost of catalytic performance in thiol–yne click reaction enabled by a palladium diketonate complex with a hexafluoroacetylacetonate ligand - Catalysis Science & Technology (RSC Publishing)
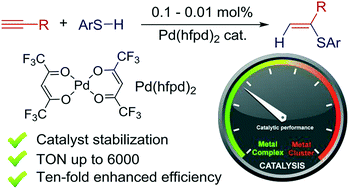
Ten-fold boost of catalytic performance in thiol–yne click reaction enabled by a palladium diketonate complex with a hexafluoroacetylacetonate ligand - Catalysis Science & Technology (RSC Publishing)
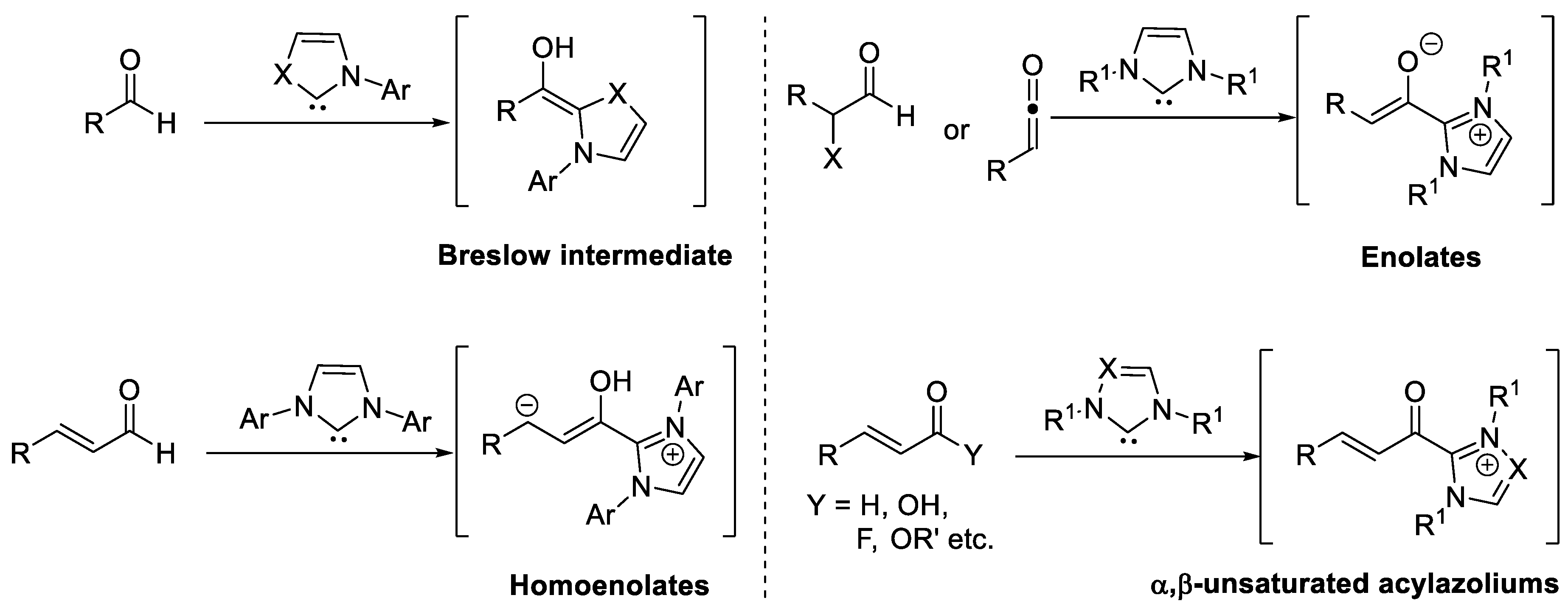
Catalysts | Free Full-Text | The Combination of Lewis Acid with N-Heterocyclic Carbene (NHC) Catalysis | HTML

Palladium/Nucleophilic Carbene Catalysts for Cross-Coupling Reactions | Johnson Matthey Technology Review

Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect
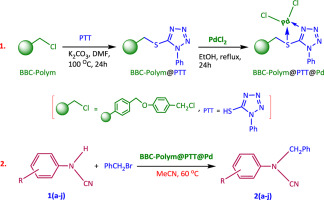
A novel tetrazole functionalized polymer-supported palladium nano-catalyst for the synthesis of various N-benzylated arylcyanamides - J. Alloys Compd. - X-MOL
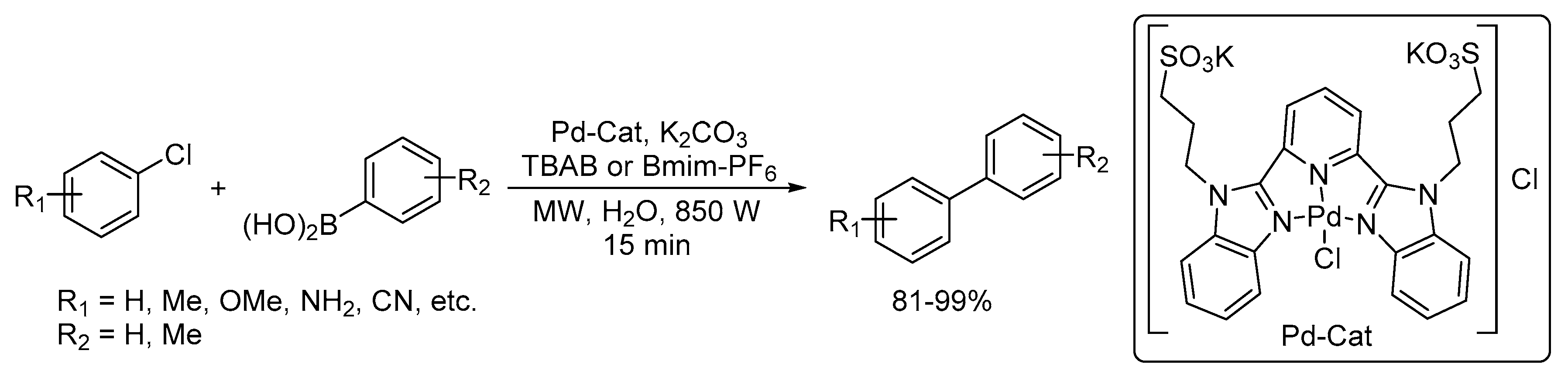
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML


![Various [NHC-Pd-PEPPSI] (left) and long alkyl chain-labeled... | Download Scientific Diagram Various [NHC-Pd-PEPPSI] (left) and long alkyl chain-labeled... | Download Scientific Diagram](https://www.researchgate.net/profile/Muhammad_Ghufran_Rafique/publication/317660984/figure/fig1/AS:528262394638337@1502958977918/Various-NHC-Pd-PEPPSI-left-and-long-alkyl-chain-labeled-NHC-Pdallyl-cinnamyl.png)