Palladium‐Catalyzed Synthesis of Benzophenanthrosilines by C−H/C−H Coupling through 1,4‐Palladium Migration/Alkene Stereoisomerization - Tsuda - - Angewandte Chemie - Wiley Online Library
Catalysts | Free Full-Text | Palladium-Catalyzed Intermolecular Oxidative Amination of Alkenes with Amines, Using Molecular Oxygen as Terminal Oxidant | HTML

Palladium(II)-catalyzed alkene functionalization via nucleopalladation: stereochemical pathways and enantioselective catalytic applications. - Abstract - Europe PMC
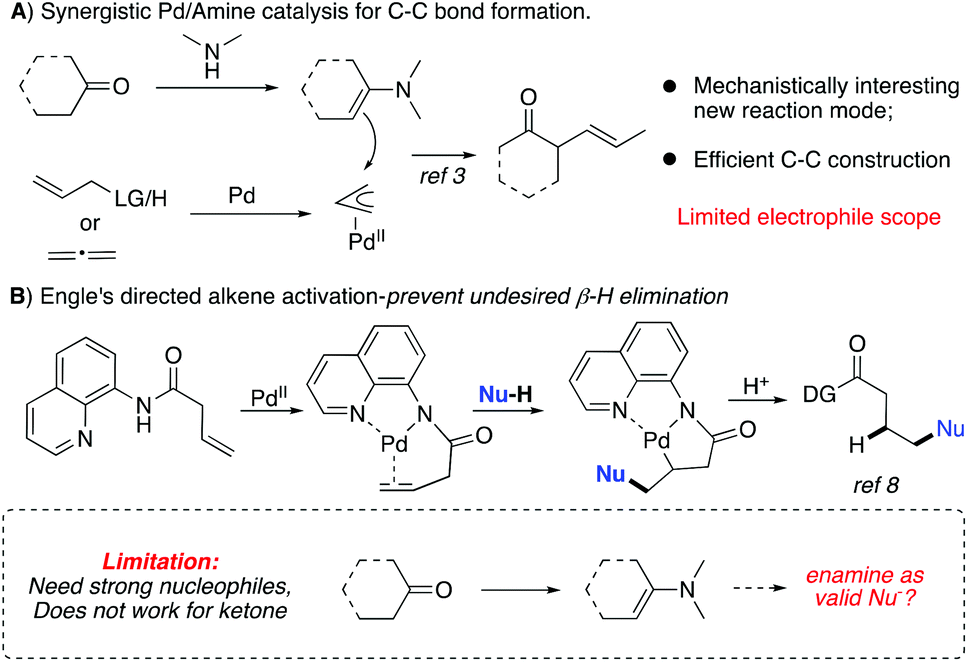
Synergistic palladium/enamine catalysis for asymmetric hydrocarbon functionalization of unactivated alkenes with ketones - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB01165J
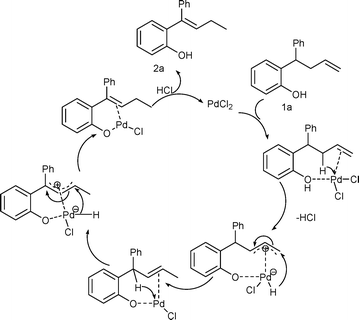
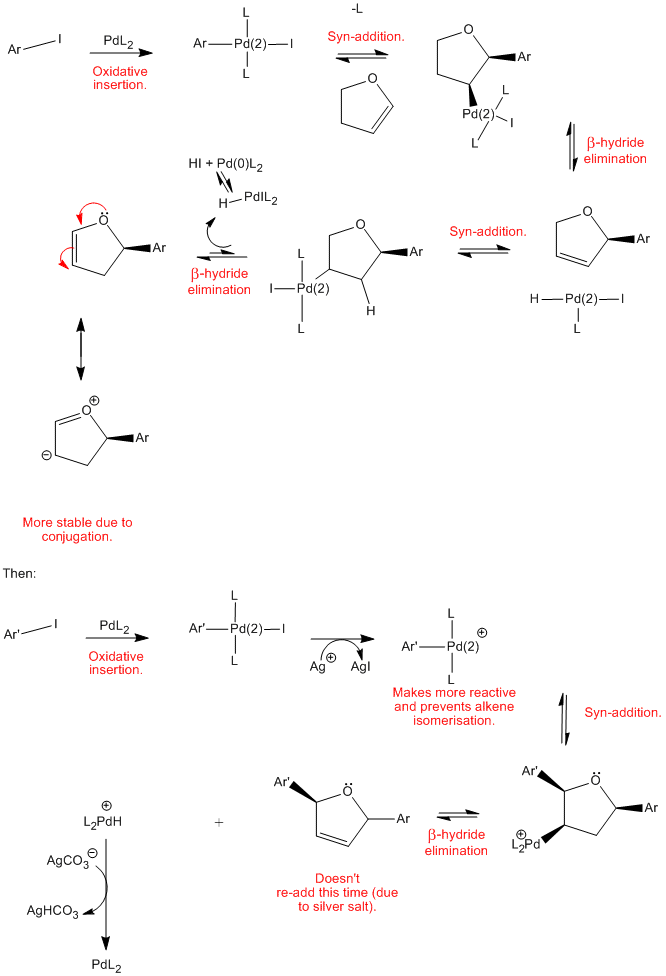
Palladium catalyzed isomerization of alkenes: a pronounced influence of an o-phenol hydroxyl group - Organic & Biomolecular Chemistry (RSC Publishing)
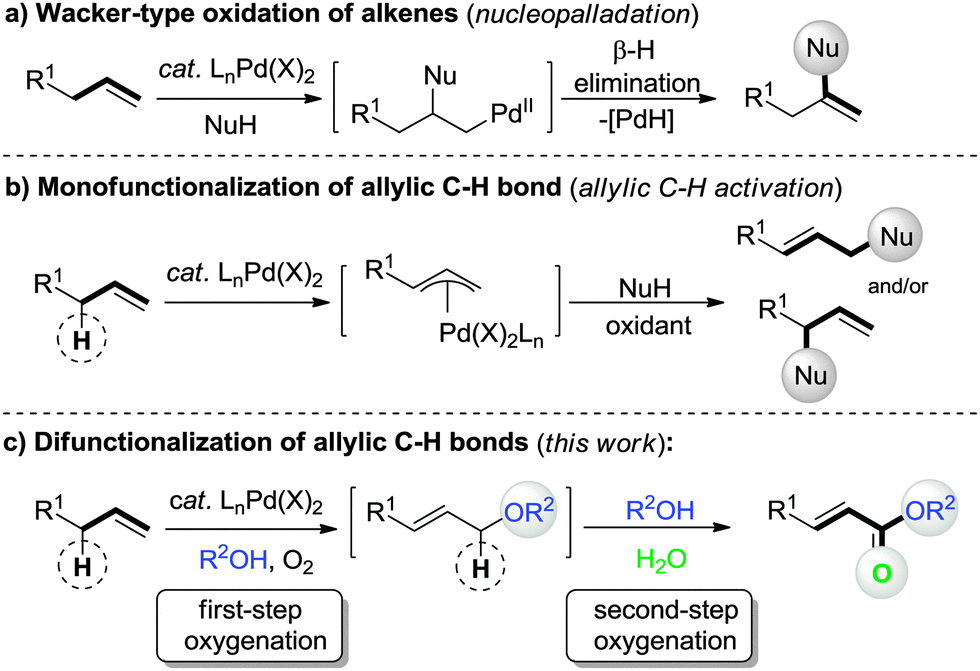
Palladium-catalyzed aerobic oxidative double allylic C–H oxygenation of alkenes: a novel and straightforward route to α,β-unsaturated esters - Chemical Communications (RSC Publishing)
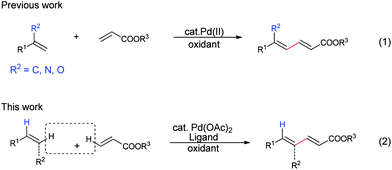
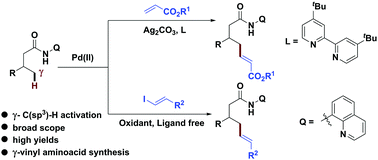
Palladium(ii)-catalyzed cross-coupling of simple alkenes with acrylates: a direct approach to 1,3-dienes through C–H activation - Chemical Science (RSC Publishing)

Figure 1 from Palladium-Catalyzed Enantioselective 1,1-Fluoroarylation of Aminoalkenes | Semantic Scholar
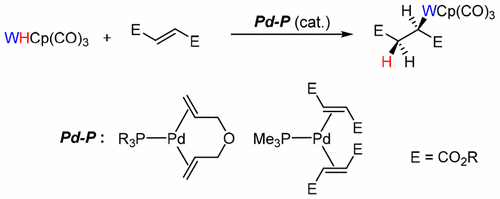
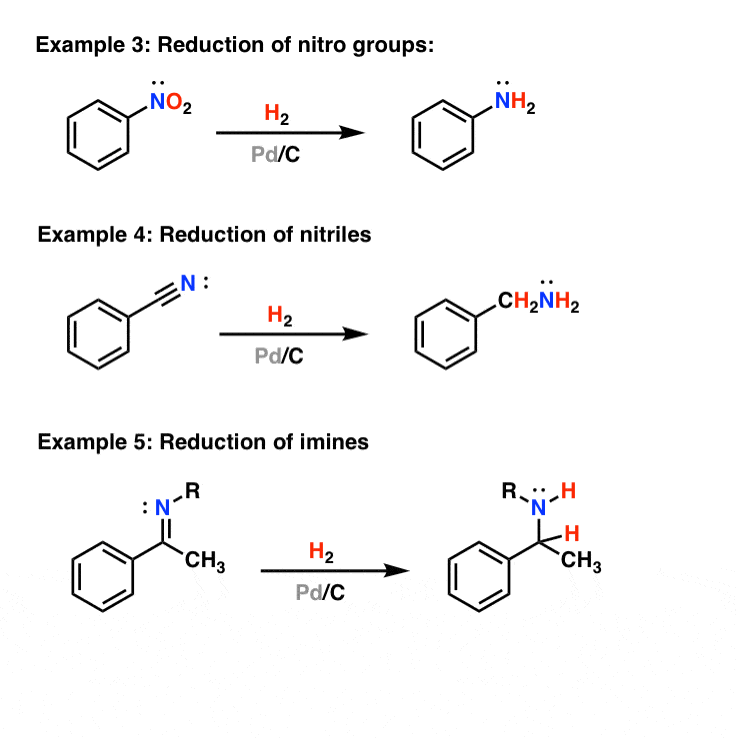
Selective Alkene Insertion into Inert Hydrogen–Metal Bonds Catalyzed by Mono(phosphorus ligand)palladium(0) Complexes - Organometallics - X-MOL
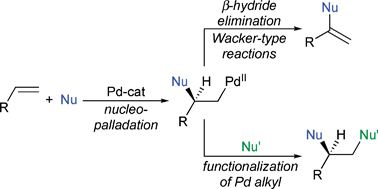
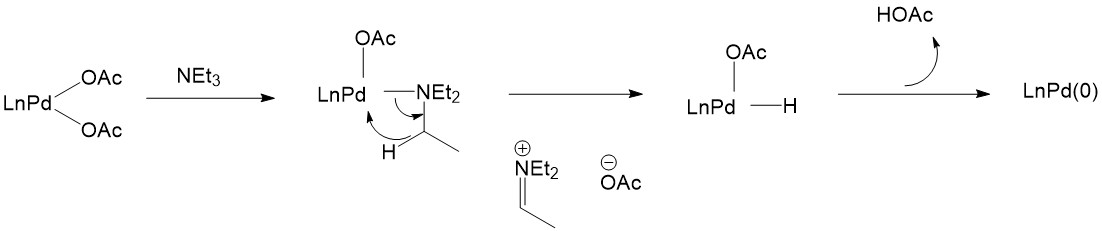
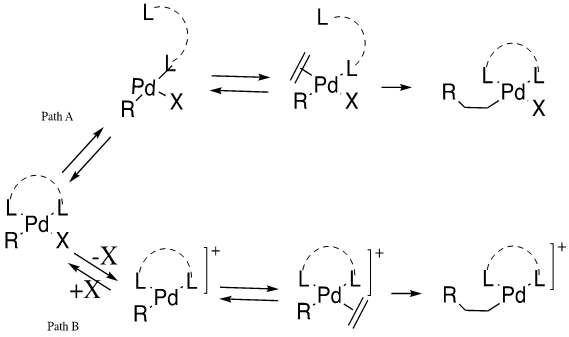
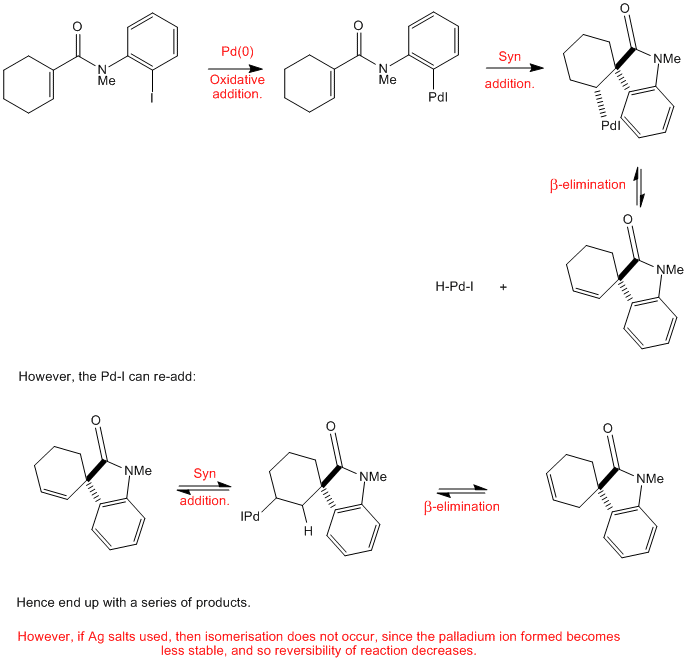
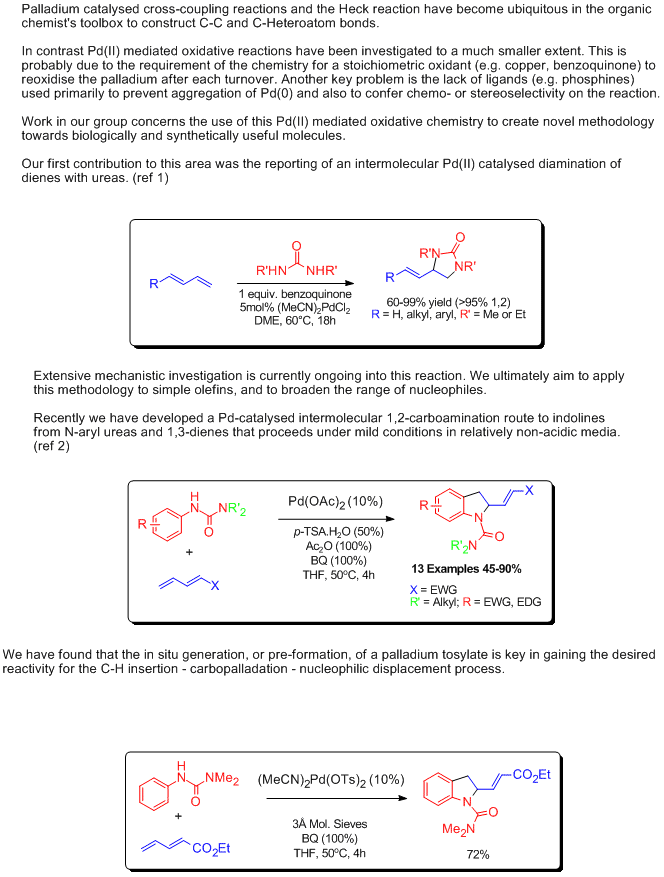
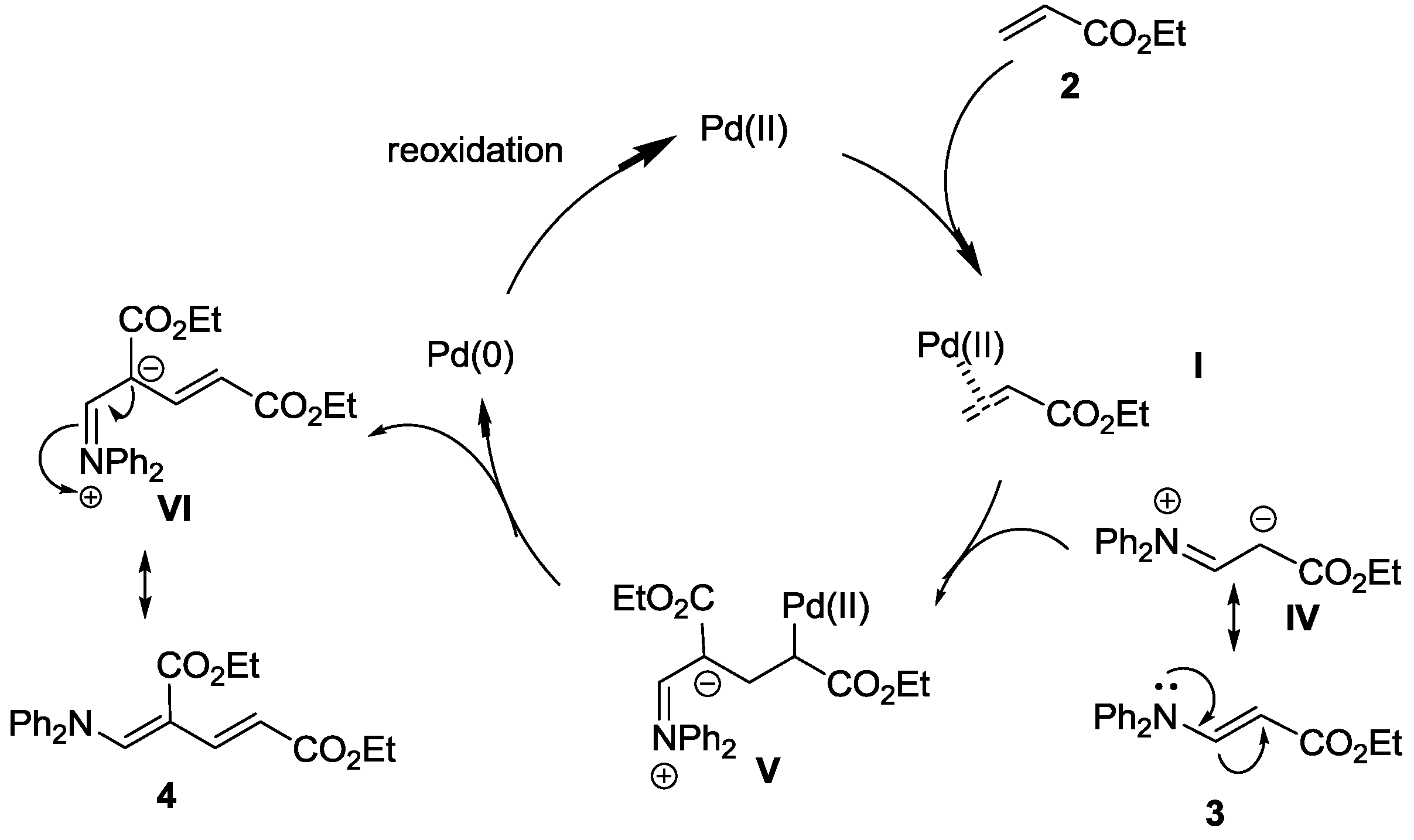
Mechanistic approaches to palladium-catalyzed alkene difunctionalization reactions - Organic & Biomolecular Chemistry (RSC Publishing)