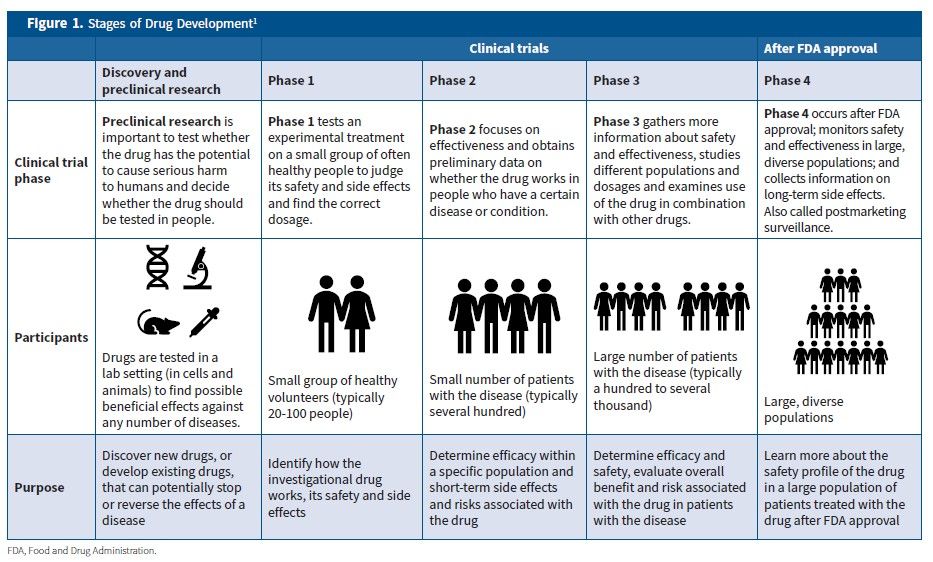
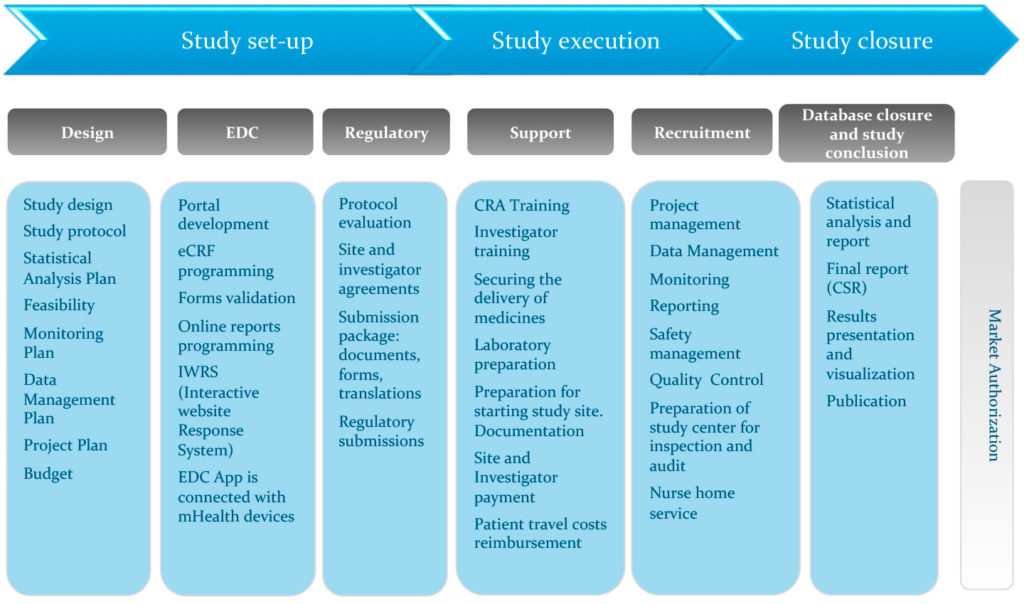
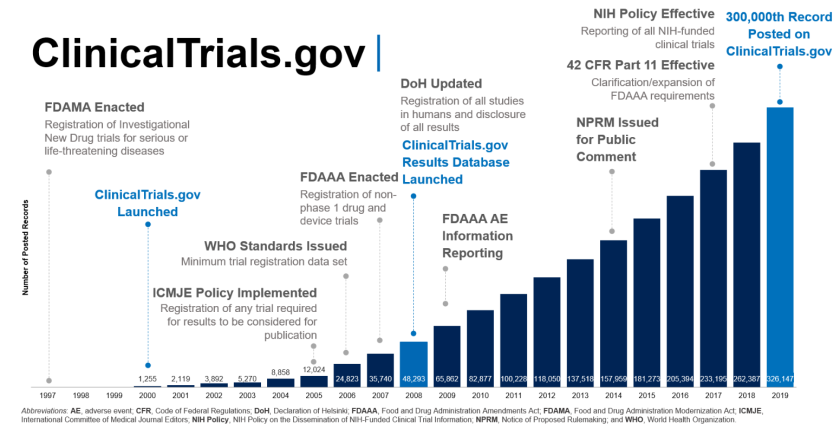
Final Rule Confirms, Posting of Study Results on ClinicalTrials.gov Will be Required for Unapproved Products - IMPACT Pharmaceutical Services, Inc.

Clinical Trials - Grey Literature in the Health Sciences - Research Guides at University of Alabama - Birmingham

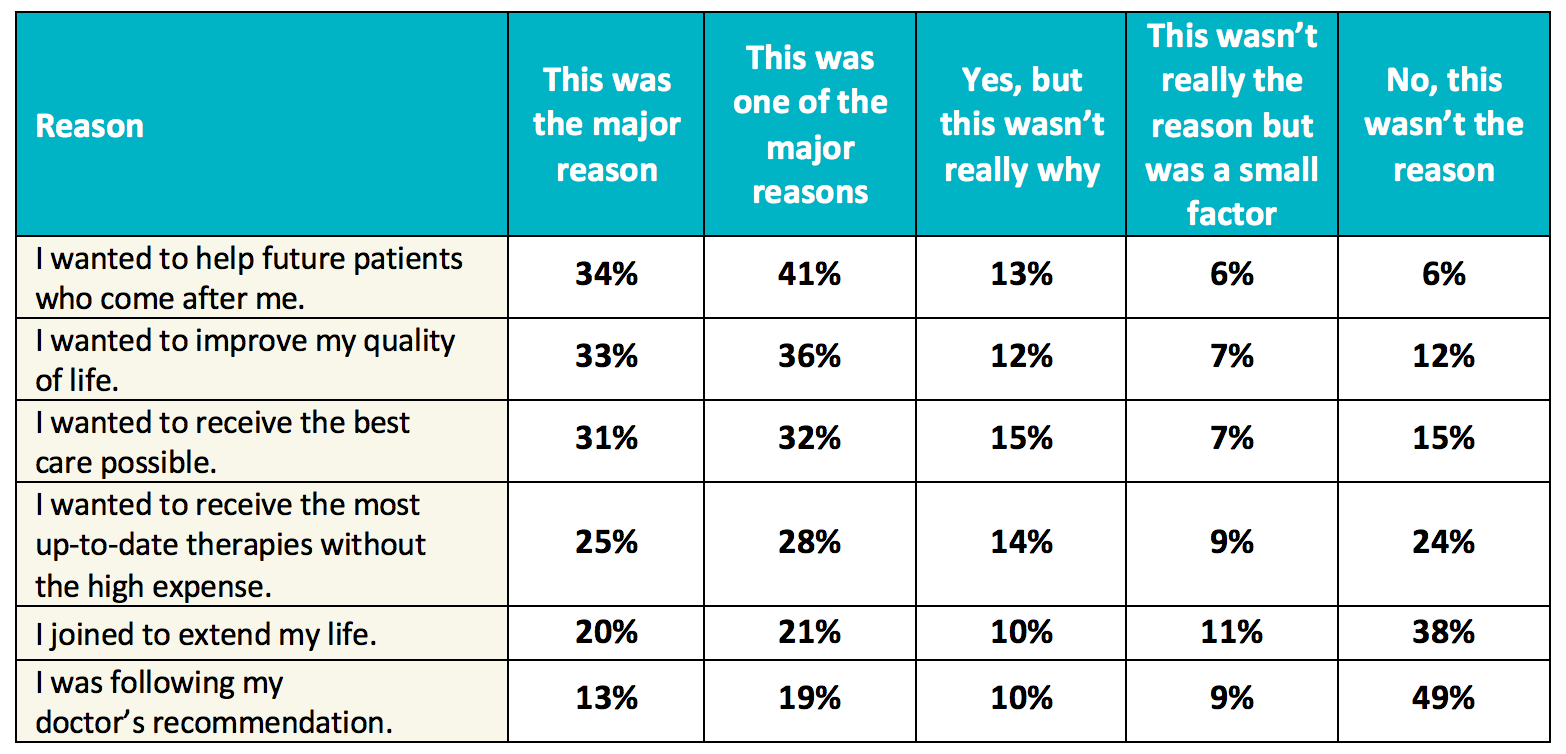
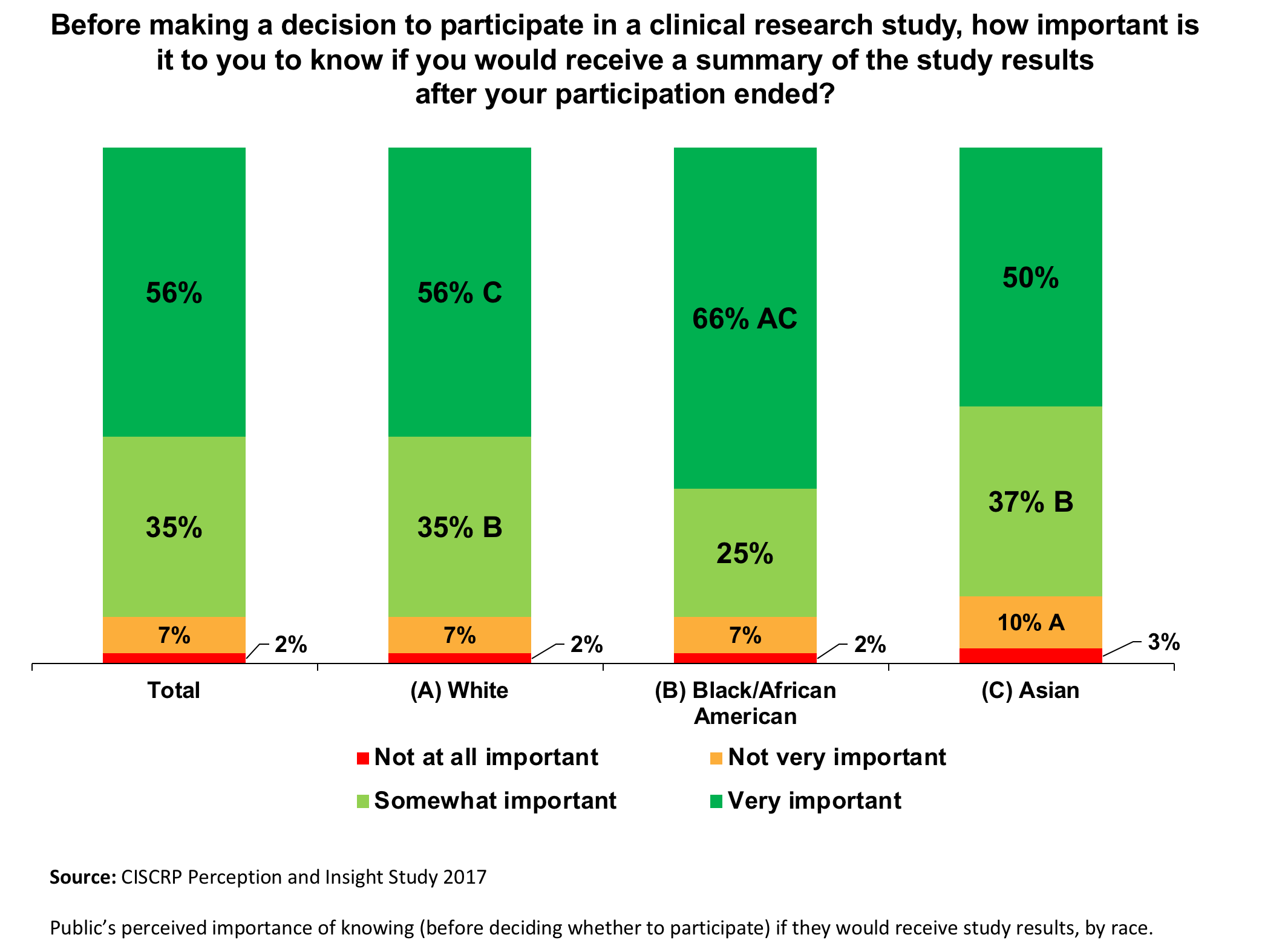
A Guide to Understanding Clinical Trials: Part 2 - Five Factors to Consider When Evaluating Results - Bench Press
How Frequently Do the Results from Completed US Clinical Trials Enter the Public Domain? - A Statistical Analysis of the ClinicalTrials.gov Database | PLOS ONE
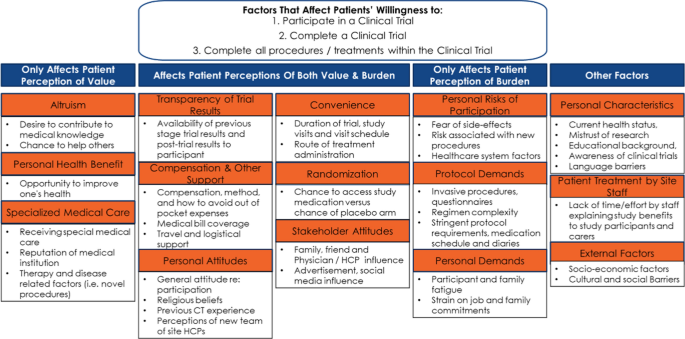
Amplifying the Voice of the Patient in Clinical Research: Development of Toolkits for Use in Designing and Conducting Patient-Centered Clinical Studies | SpringerLink
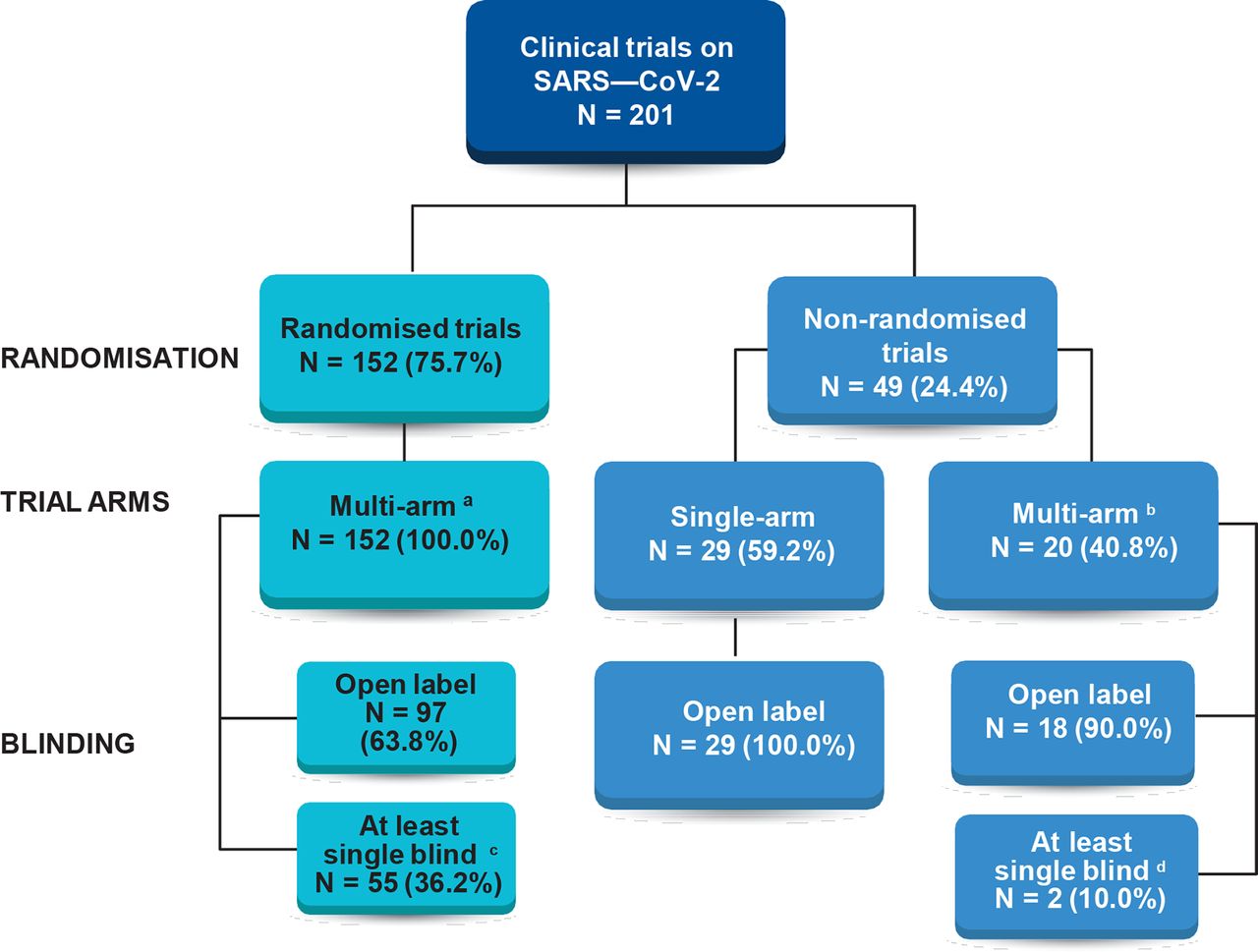
Characteristics of registered clinical trials assessing treatments for COVID-19: a cross-sectional analysis | BMJ Open